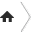Marble is a nonfoliated metamorphic rock resulting from the metamorphism of limestone, composed mostly of calcite (a crystalline form of calcium carbonate, CaCO3). It is extensively used for sculpture, as a building material, and in many other applications. The word "marble" is colloquially used to refer to many other stones that are capable of taking a high polish.
The word "marble" derives from the Greek μάρμαρον (marmaron)[1] and that from μάρμαρος (marmaros), "crystalline rock", "shining stone"[2][3], perhaps from the verb μαρμαίρω (marmairō), "to flash, sparkle, gleam"[4]. This stem is also the basis for the English word marmoreal, meaning "marble-like".
Origins
Marble is a metamorphic rock resulting from regional or rarely contact metamorphism of sedimentary carbonate rocks, either limestone or dolomite rock, or metamorphism of older marble. This metamorphic process causes a complete recrystallization of the original rock into an interlocking mosaic of calcite, aragonite and/or dolomite crystals. The temperatures and pressures necessary to form marble usually destroy any fossils and sedimentary textures present in the original rock.
Pure white marble is the result of metamorphism of very pure limestones. The characteristic swirls and veins of many colored marble varieties are usually due to various mineral impurities such as clay, silt, sand, iron oxides, or chert which were originally present as grains or layers in the limestone. Green coloration is often due to serpentine resulting from originally high magnesium limestone or dolostone with silica impurities. These various impurities have been mobilized and recrystallized by the intense pressure and heat of the metamorphism.
Construction Marble
In the construction, specifically the dimension stone trade, the term "marble" is used for any crystalline calcitic rock (and some non-calcitic rocks) useful as building stone. For example, "Tennessee marble" is really a dense granular fossiliferous gray to pink to maroon Ordovician limestone that geologists call the Holston Formation.
Industrial use of marble
Colorless or light-colored marbles are a very pure source of calcium carbonate, which is used in a wide variety of industries. Finely ground marble or calcium carbonate powder is a component in paper, and in consumer products such as toothpaste, plastics, and paints. Ground calcium carbonate can be made from limestone, chalk, and marble; about three-quarters of the ground calcium carbonate worldwide is made from marble. Ground calcium carbonate is used as a coating pigment for paper because of its high brightness and as a paper filler because it strengthens the sheet and imparts high brightness. Ground calcium carbonate is used in consumer products such as a food additive, in toothpaste, and as an inert filler in pills. It is used in plastics because it imparts stiffness, impact strength, dimensional stability, and thermal conductivity. It is used in paints because it is a good filler and extender, has high brightness, and is weather resistant. However, the growth in demand for ground calcium carbonate in the last decade has mostly been for a coating pigment in paper.
Calcium carbonate can also be reduced under high heat to calcium oxide (also known as "lime"), which has many applications including being a primary component of many forms of cement.
From: Wikipedia